- "Students are given the opportunity to assess chemical products and processes and design greener alternatives when appropriate."
- "Students understand and can evaluate the environmental, social, and health impacts of a chemical product over the life cycle of the product, from synthesis to disposal."
At the highest level of the ACS guidelines, students are expected to have a deeper understanding of and critical thinking skills around developing and designing greener chemical products and processes. This can include the various environmental, social and health aspects of a chemical product, in particular as it pertains to life cycle analysis.
GCTLC Library
Below are resources from the GCTLC library that are tagged with any of the following Green Chemistry Principles:
- #3 (Less Hazardous Chemical Syntheses)
- #4 (Design Safer Chemicals)
- #5 (Safer Solvents and Auxiliaries)
OR have been tagged with keyword "Life Cycle Assessment".
The variety of resources below should provide educators with numerous options to help tailor their lectures and courses with more green and sustainable chemistry content. However, if you have additional suggestions for resources, you can always submit them for inclusion in the GCTLC library, or you can post them in the forum "Green Chemistry Resources for Addressing the ACS Guidelines" on the GCTLC.
A Green Alternative to Aluminum Chloride Alkylation of Xylene
The alkylation of aromatic compounds is a very important industrial process. Typically these alkylations are performed using strong Lewis acids like aluminum trichloride as catalysts. However, the use of these catalysts often requires an aqueous work-up to remove the aluminum and also leads to mixtures of products due to carbocation rearrangement.
By using graphite this greener alkylation ...

A Green and Efficient Cyclization of Citronellal into Isopulegol: A Guided-Inquiry Organic Chemistry Laboratory Experiment
Citronellal is a naturally occurring aldehyde derived from essential oils like citronella. In this laboratory activity, citronellal undergoes a cyclization reaction to form isopulegol, a chiral terpene with various applications. The reaction utilizes montmorillonite (MK10) clay as a catalyst in a buffer medium at room temperature for 2 hours. NMR Spectroscopy is used to characterize the product. ...

A Green Nucleophilic Aromatic Substitution Reaction
This laboratory activity focuses on the tie-dyeing process as a safer alternative to nucleophilic aromatic substitution (SNAr) reactions for an introductory organic chemistry laboratory. The simple and straightforward experiment provides students with an opportunity to gain practical experience in conducting a chemical reaction in a real-world context while applying concepts of design for ...
A Green, Guided-Inquiry Based Electrophilic Aromatic Substitution for the Organic Chemistry Laboratory
This exercise demonstrates the regioselectivity of substituted aromatic systems while reducing the amount and hazards of the waste produced by the class. Although typical iodinations involve either high amounts of derivatization (i.e., nitration, then reduction to aniline, formation of diazonium, then ionization) or use of hazardous/expensive reagents (iodine and mercury acetate, bis(pyridine ...
A Greener Approach for Measuring Colligative Properties
This experiment teaches students the concept of colligative properties using renewable, benign, and biodegradable materials. Rather than using conventional solvents with high cryoscopic constants (which are oftentimes aromatics), fatty acids are used to explore freezing-point depression. By monitoring the change in freezing points of solutions with varying solute concentrations, students will be ...
A Greener Chemiluminescence Demonstration
This demonstration shows students a long-lasting, chemiluminescent reaction in light sticks without the toxic solvents. The instructor must prepare the chemiluminescent reagent, divanillyl oxalate, in a one-step synthetic procedure or can have the students prepare the reagent as part of an organic chemistry experiment.
Solvents acceptable for the chemiluminescence reaction are ethyl acetate or ...

A Guide To Green Chemistry Experiments for Undergraduate Organic Chemistry Labs
Beyond Benign, My Green Lab, and MilliporeSigma have teamed up to develop a comprehensive teaching guide for undergraduate labs featuring Green Chemistry alternatives to traditional organic chemistry experiments and Green Chemistry lab practices*.
The purpose of this guide is to provide examples of green chemistry experiments that may serve as substitutions for classic undergraduate organic ...

A guidebook for sustainability in laboratories
This guidebook aims to improve lab users’ everyday practices to become more sustainable. Specifically, this guidebook provides practical suggestions on how to effectively use lab instruments and resources and how to acquire data. We provide advice to labs covering disciplines such as biology, chemistry, computational science, engineering, life sciences, materials sciences, medicine, pharmacy, and ...
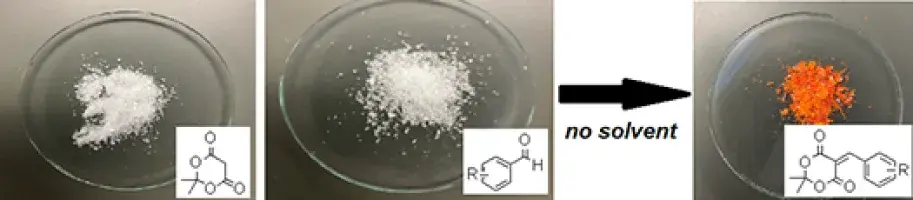
A Guided Inquiry Laboratory Activity to Explore Reactivity of Carbonyl Compounds
A laboratory activity to demonstrate reactivity of carbonyl compounds using Meldrum's acid as a nucleophile is presented. The experiment requires no solvent and can be completed in a single 3-4 hour laboratory period. Substrate information and related spectroscopic information are provided.
Tracey, M. P., Nigam, M., Pirzada, E., & Osman, T. (2024). A solventless carbonyl addition reaction as a ...

A Microscale Heck Reaction In Water
This laboratory experiment features the palladium-catalyzed Heck synthesis of (E)-4-acetylcinnamic acid from 4-iodoacetophenone and acrylic acid by mid-level undergraduates. Traditional Heck reaction organic solvents (e.g. acetonitrile) and base (e.g. triethylamine) are replaced by water and sodium carbonate respectively. This approach introduces fundamentals of green chemistry (aqueous and ...
Pagination
- Previous page
- Page 3
- Next page