Bioleaching of Rare Earth Fluorescent Lamp Phosphors Using Kombucha
Summary
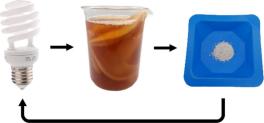
Rare earth elements (REEs) are critical for modern technologies but are rarely recycled, with less than 1% recovered from waste products. Yttrium, a key component in fluorescent lamps and light emitting diodes, poses particular recycling challenges despite its high concentration in such materials. Here, we present an innovative bioleaching approach for yttrium recovery using the fermented tea beverage Kombucha in the context of science communication and outreach. Kombucha contains organic acid producing microorganisms, which can mobilize yttrium from a model phosphor mixture. In this experiment, high school students prepare Kombucha, mix it with phosphor powder, and allow bioleaching to occur over 7 to 14 days. The dissolved yttrium is then precipitated as yttrium oxalate using oxalic acid and thermally converted back to yttrium oxide. Yields of yttrium oxide ranged from 7% to 13%, depending on the maturity of the Kombucha culture and the experimental conditions. This model experiment enables students to understand bioleaching as a sustainable alternative to conventional chemical extraction, illustrating its advantages such as reduced environmental impact and energy use. Furthermore, it highlights the potential of bioleaching to improve the recycling of critical materials such as yttrium, aligning with circular economy goals.
Full citation: Fritz, A.P.; Daumann, L.J.; Schwarzer, S. J. Chem. Educ. 2025, DOI: https://doi.org/10.1021/acs.jchemed.4c01532
Full citation: Fritz, A.P.; Daumann, L.J.; Schwarzer, S. J. Chem. Educ. 2025, DOI: https://doi.org/10.1021/acs.jchemed.4c01532
Link to external
Creative Commons License
